2025 AGA Distinguished Achievement Award in Basic Science to Eugene B. Chang, MD, AGAF
One challenge of this millennium is dealing with the onslaught of ΓÇ£new ageΓÇ¥ complex immune disorders like inflammatory bowel diseases, type 1 diabetes, Celiac disease, asthma, and rheumatoid arthritis, as well as more common diseases such as colon cancer, type 2 diabetes, and obesity. The insidious increase in these diseases over the past several decades is likely due to shifts in environmental factors and societal norms brought on by cultural Westernization. Accompanying these are dramatic changes in the collective human microbiome of the gastrointestinal tract, a community of trillions of microbial organisms that together function as an ΓÇ£acquiredΓÇ¥ organ of our bodies, essential to sustaining health.
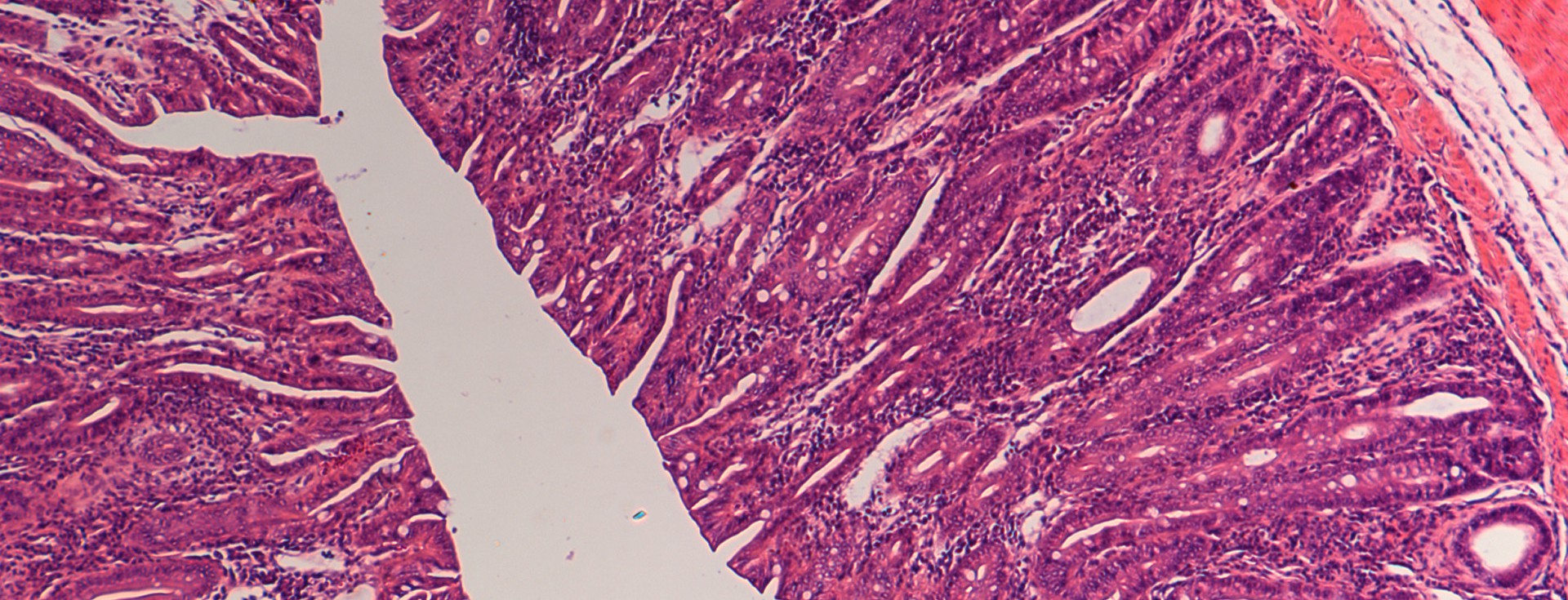
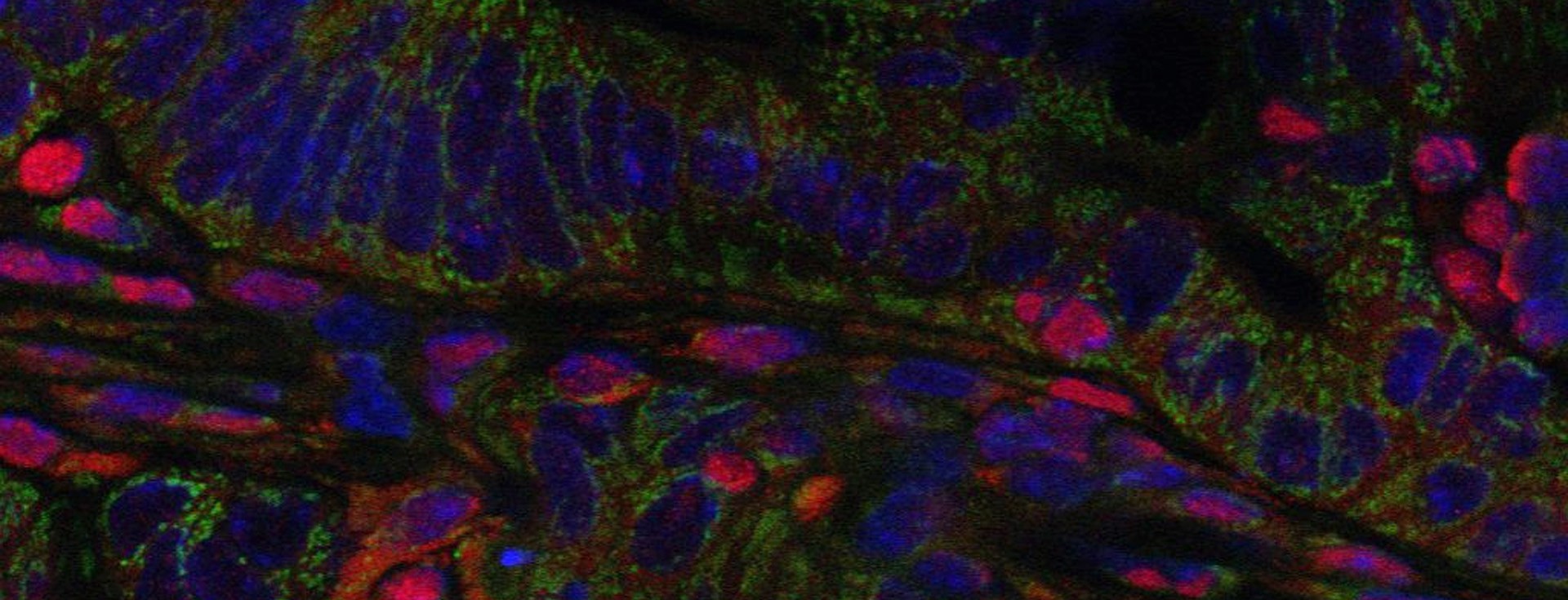
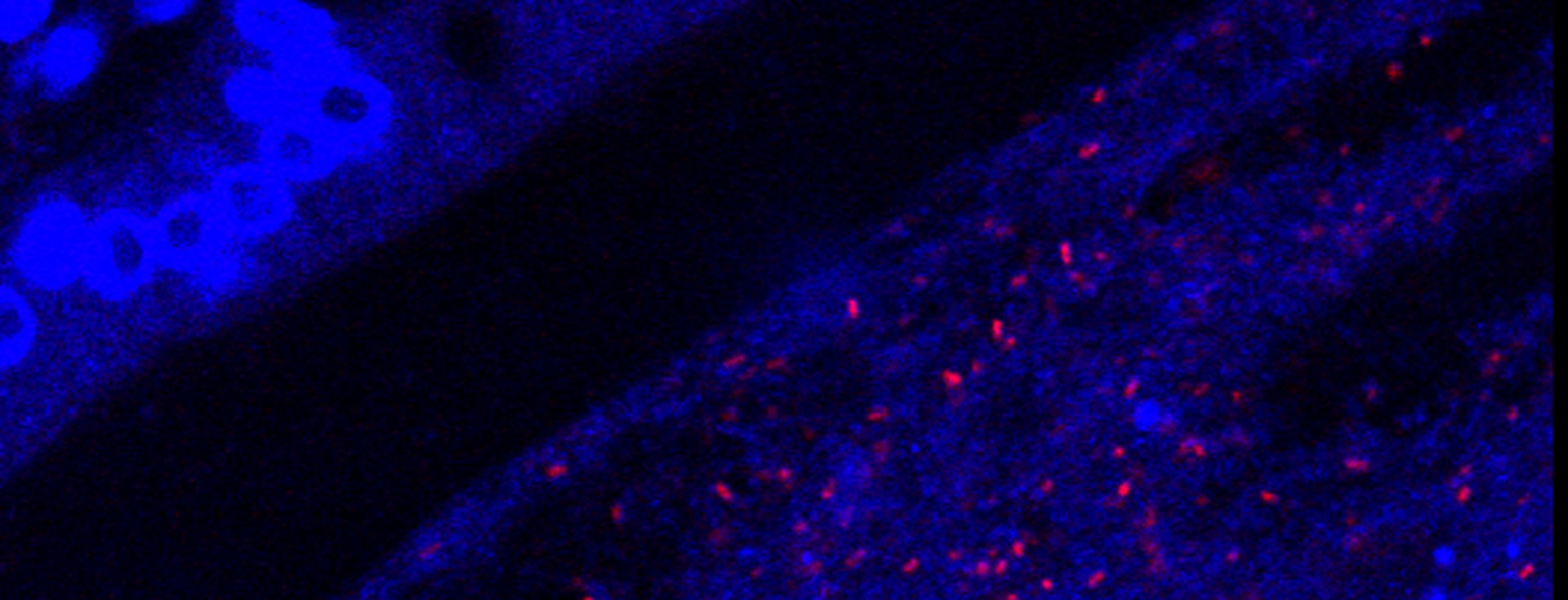
The relationship of the intestinal microbiota and the human host is fundamental to our health and, when perturbed or dysbiosis occurs, the consequences can be catastrophic. In genetically susceptible individuals, these factors can potentially trigger events that disturb immune and metabolic homeostasis, initiating the development of disease.
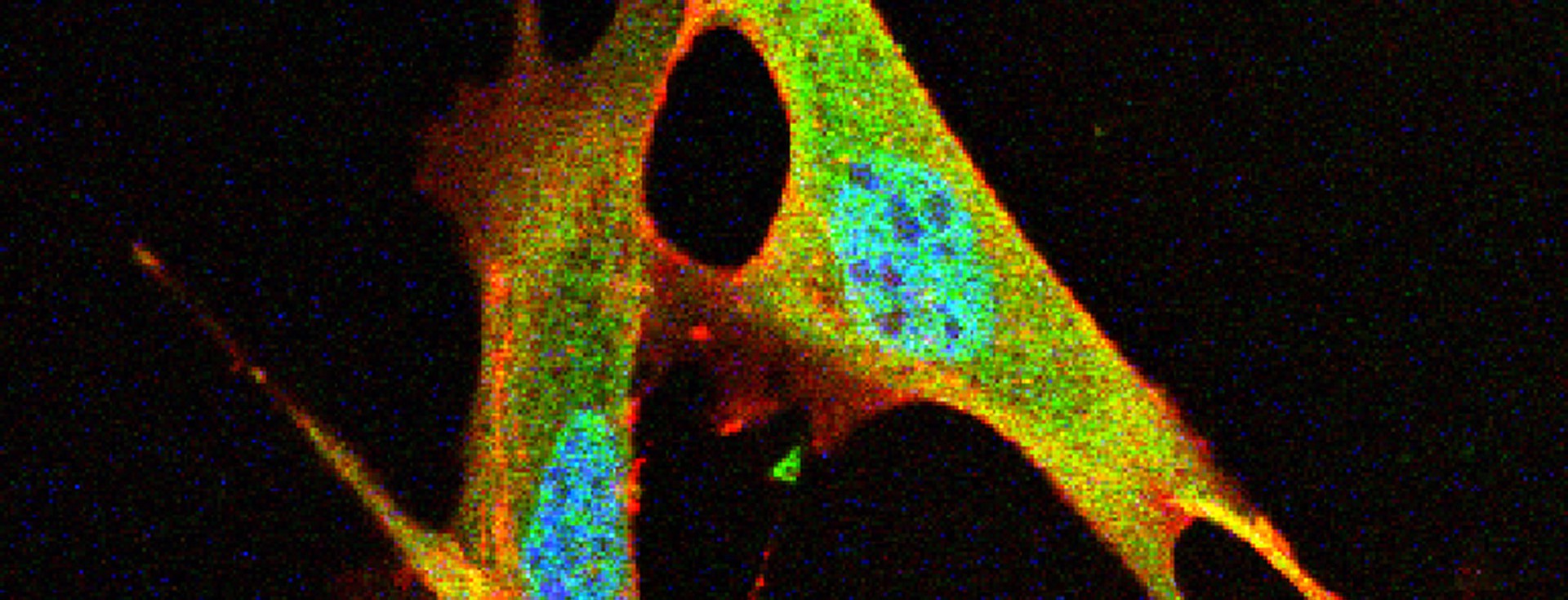
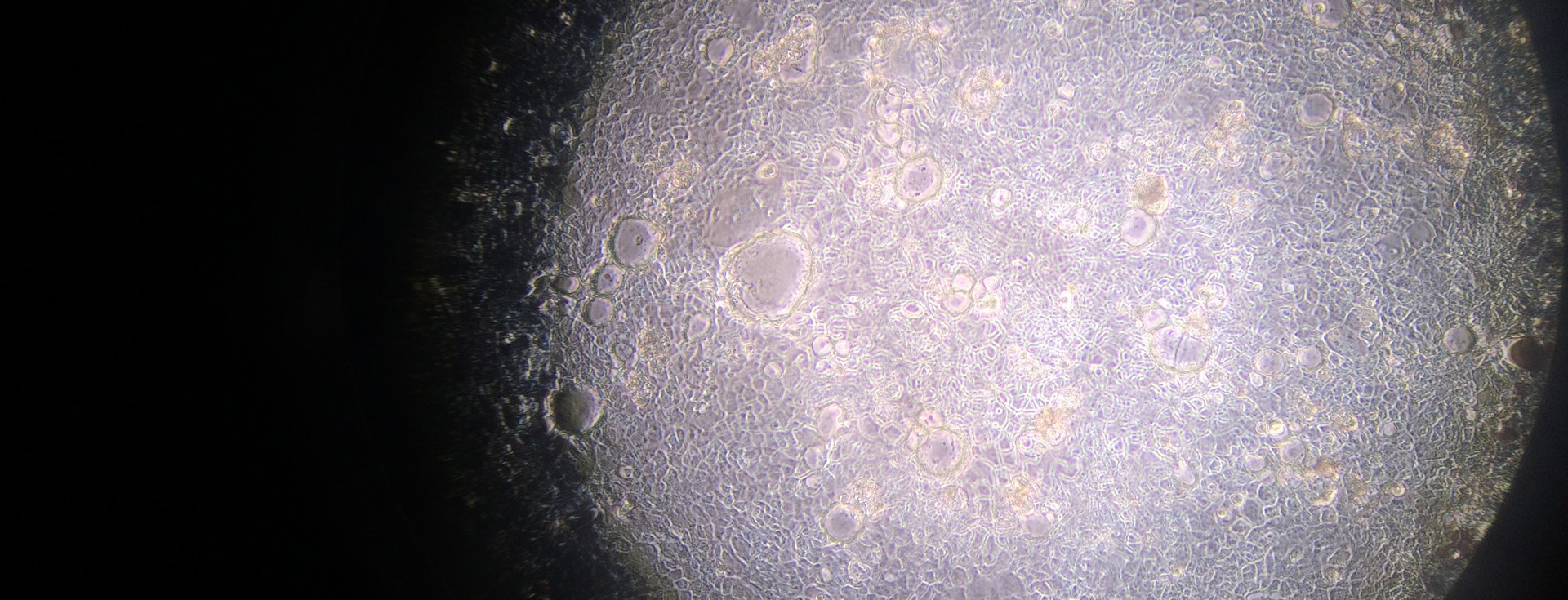
Our efforts are therefore directed towards gaining a better understanding of what factors are involved in the selection and assembly of intestinal microbes, and how they can be used to reshape the enteric microbiome to prevent and treat disease. The focus of my group is the study of host-microbe interactions in the context of human and mammalian systems and the bidirectional signaling processes that mediate these interactions. We employ cutting edge approaches that include cultivation-dependent and ΓÇôindependent technologies for microbial analysis, genetically modified and gnotobiotic mouse models, metabolic and functional measurements, and advanced bioinformatic tools to investigate both the host and the microbiome in order to address these questions.